Unsere Schwerpunkte: Volkserkrankungen wie Asthma, Allergien und Chronisch Obstruktive Lungenerkrankungen (COPD), sowie infektionsbedingte Entzündungen der Lunge, vor allem die Tuberkulose (TB).
14.04.2026
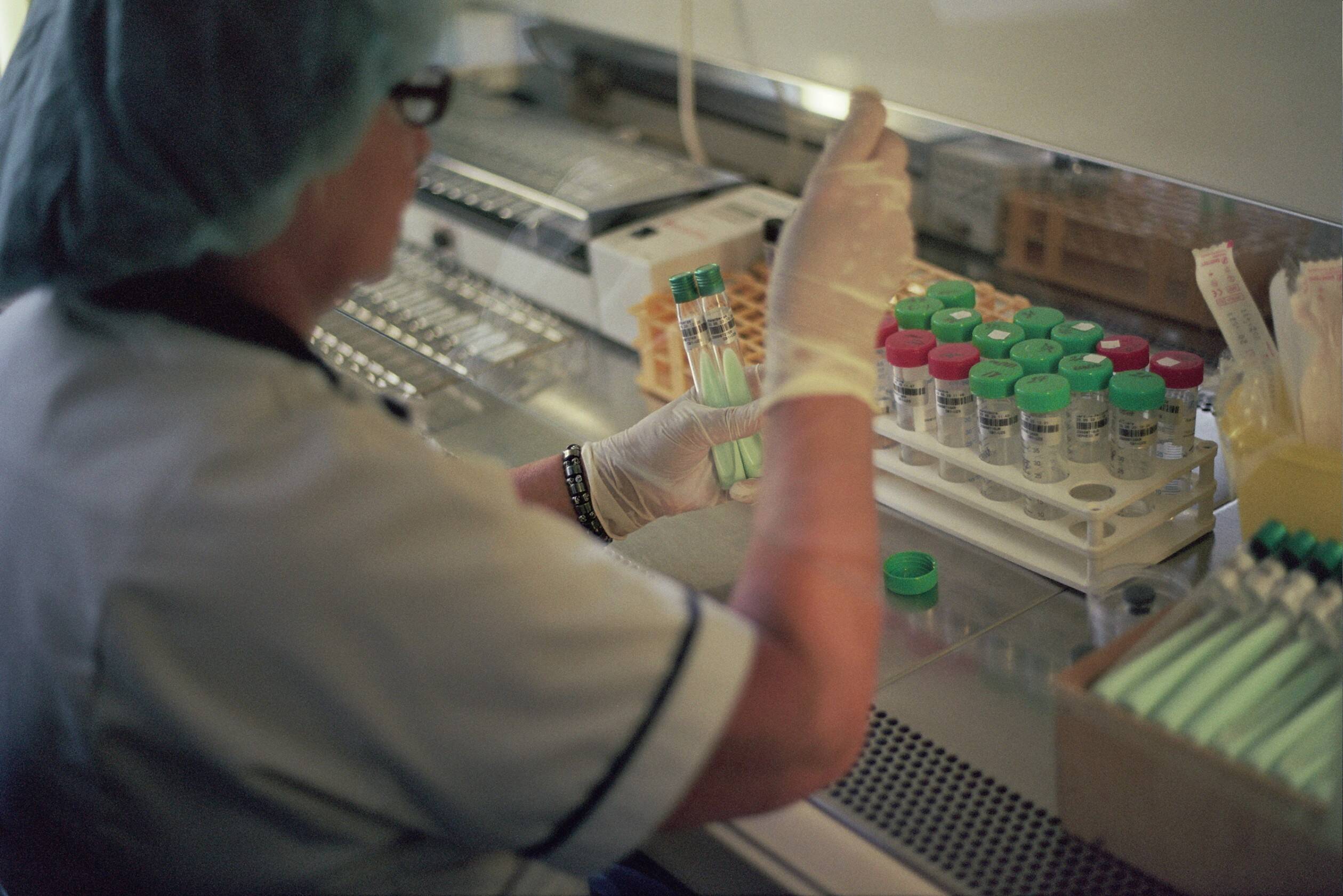
Eine neue nationale Kohortenstudie aus Lettland in Zusammenarbeit mit Foschenden der klinischen Tuberkulose-Infrastruktur (ClinTB) am Deutsche Zentrum für Infektionsforschung (DZIF) am Forschungszentrum Borstel, Leibniz Lungenzentrum (FZB), liefert wichtige Erkenntnisse zur Behandlung multiresistenter Tuberkulose (MDR-TB).

10.04.2026
Dr. Eva-Maria Rick aus der Forschungsgruppe „Klinische und Molekulare Allergologie“ (Ltg. Prof. U. Jappe) ist auf der Wissenschaftlichen Frühjahrstagung der DGAKI 2026, die vom 27.–28. März in Mainz stattfand, mit einem Abstractpreis ausgezeichnet worden.

07.04.2026
"Vorsicht, Zecke: Wenn ein Stich eine Fleischallergie auslöst" ist der Titel des Beitrages, der heute Abend um 20.15 Uhr bei VISITE - dem Gesundheitsmagazin des NDRs läuft. Prof. Uta Jappe, Leiterin der Forschungsgruppe "Klinische und Molekulare Allergologie" am FZB wird dort als Expertin zum Alpha-Gal-Syndrom interviewt.